Atlas maps
Brain regional BPND values were compared to the corresponding receptor density measurements from post-mortem autoradiography data from Varnäs et al. (2004) and Bonaventure et al. (2000) (for 5-HT4R). For all five targets, we found good to excellent associations between BPND and Bmax. The slope estimates of the regression were used to transform the BPND atlases into Bmax atlases (Figure 1 and 2), allowing for a direct comparison across targets. The regional densities are presented in Figure 3. No global or regional significant effect of age, gender or age x gender was found.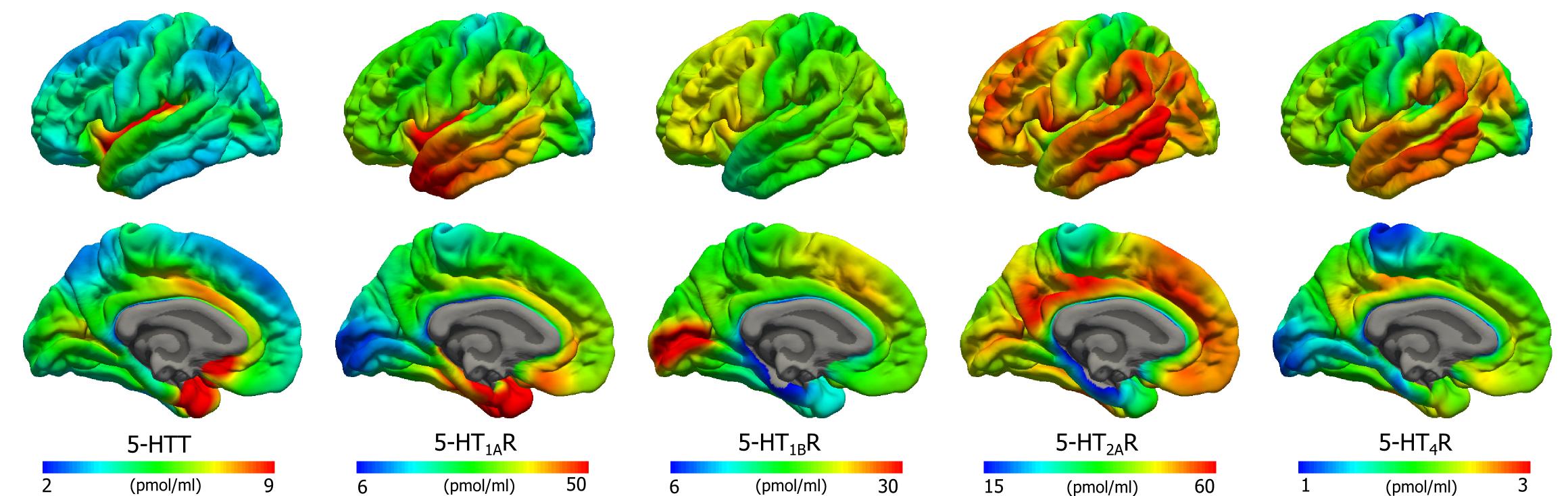
Figure 1. Average density (Bmax) maps for five 5-HT targets on the common FreeSurfer surface (left hemisphere; lateral view, upper and medial view, lower). Color scaling was individually adjusted in order to highlight features of the distributions.
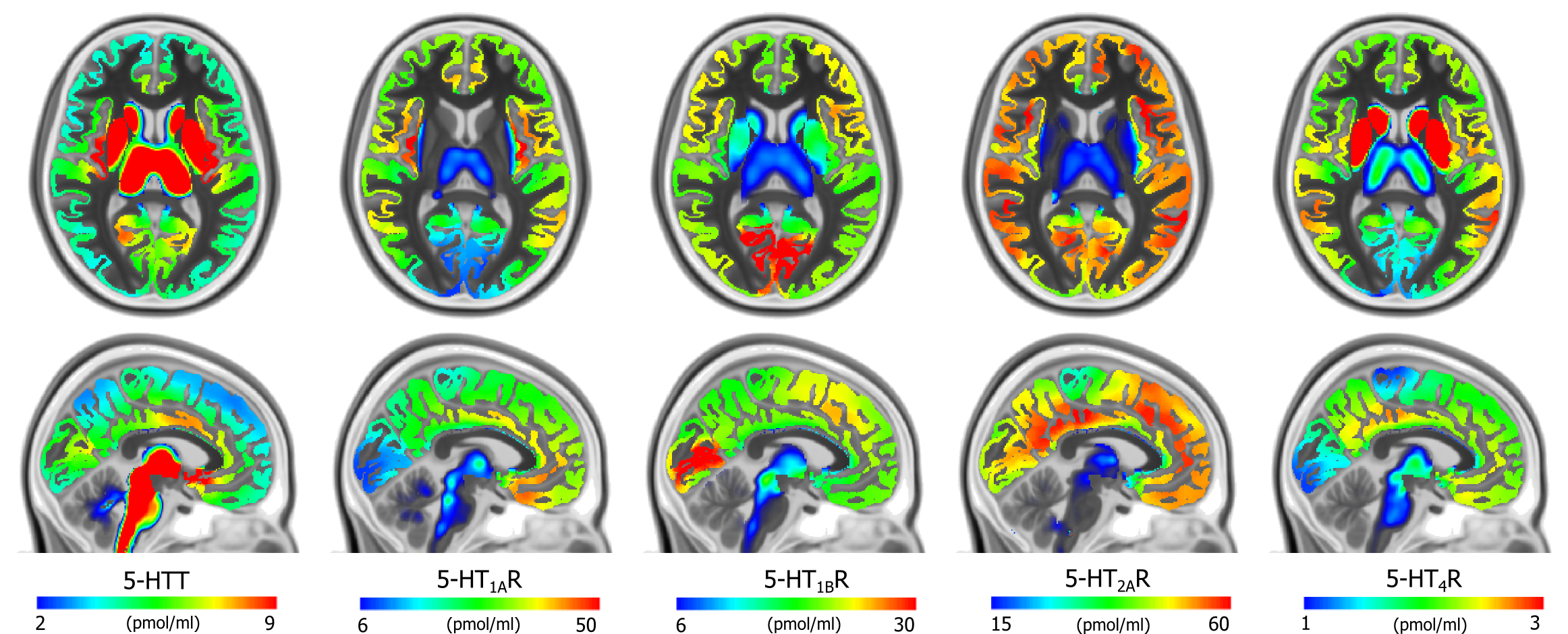
Figure 2.Average density (Bmax) maps for the five 5-HT targets in the common MNI152 space (coronal, upper, z=8mm and sagittal, lower, x=-3mm). Color scaling was individually adjusted to highlight features of the distributions.
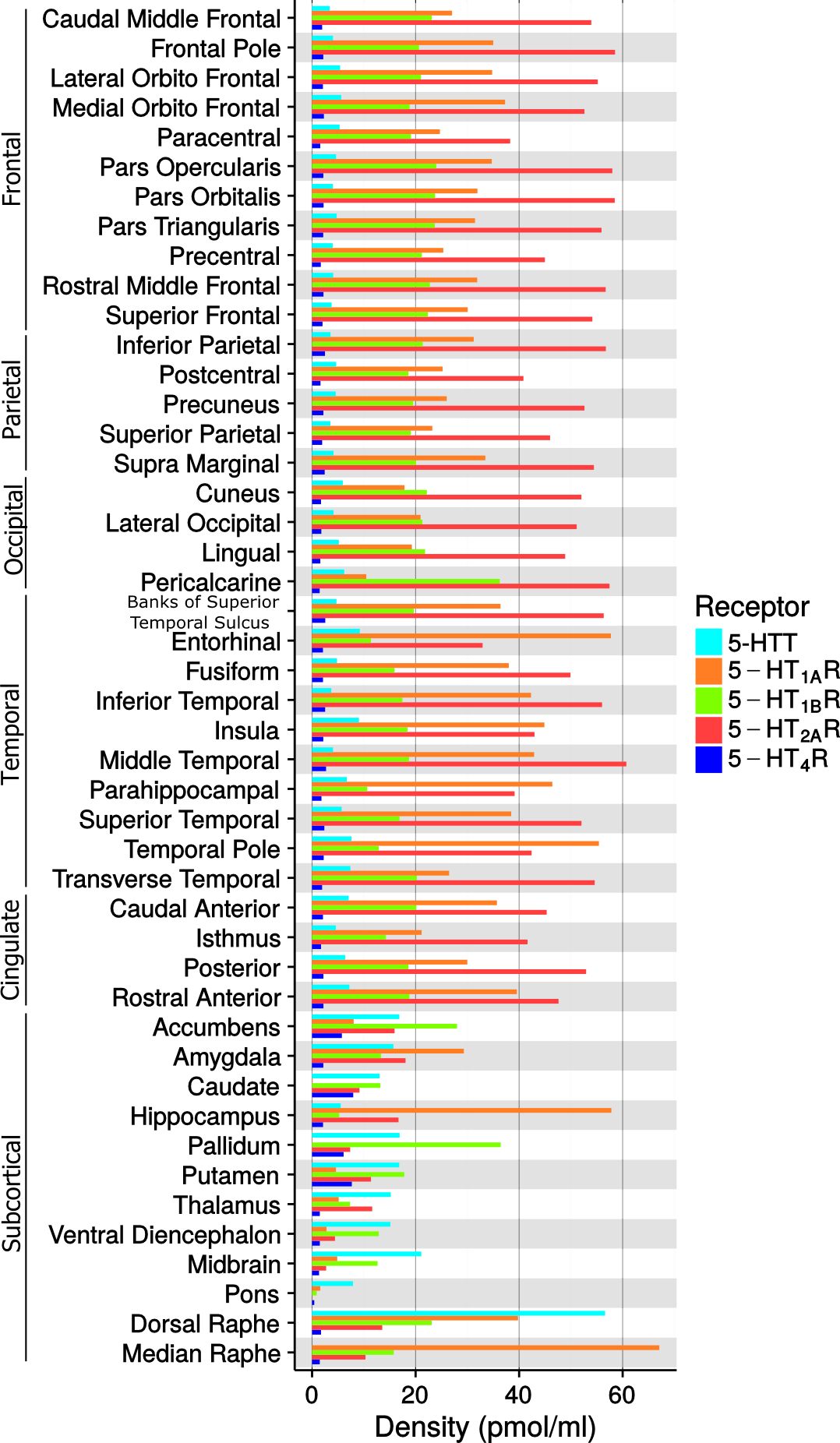
Figure 3. Density values (Bmax) of the five 5-HT targets in FreeSurfer defined brain regions. Median raphe is not reported for 5-HTT due the irreversible kinetic of the TACs, see also the Material and Methods section.
Clustering of atlas maps
The above mentioned human brain atlas of the serotonin (5-HT) system does not conform with commonly used parcellations of neocortex, since the spatial distribution of homogeneous 5-HT receptors and transporter is not aligned with such brain regions. This discrepancy indicates that a neocortical parcellation specific to the 5-HT system is needed. Hence we present parcellations of the 5-HT system created using a clustering approach focused on identifying stable and homogeneous clusters and derived from brain MR- and high-resolution PET images of five different 5-HT targets from 210 healthy controls. This is the same data that was used in the derivation of the atlas above. We then explore how well this new 5-HT parcellation can explain mRNA levels of all 5-HT genes. The parcellation derived here represents a characterization of the 5-HT system which is more stable and explains the underlying 5-HT molecular imaging data better than other atlases, and may hence be more sensitive to capture region-specific changes modulated by 5-HT.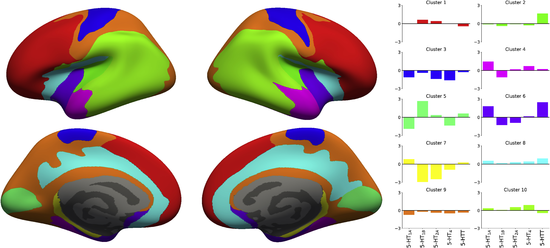
Figure 4. Parcellation obtained with K=10 clusters and the associated regional 5-HT profile for each region. The parcellation is presented on the inflated fsaverage surface medial (lower) and lateral (upper) for both hemisphere (left and right).
Publications
The following publications should be referenced when using this atlas:- Vincent Beliveau, Melanie Ganz, Ling Feng, Brice Ozenne, Liselotte Højgaard, Patrick M. Fisher, Claus Svarer, Douglas N. Greve, Gitte M. Knudsen (2017) A high-resolution in vivo atlas of the human brain's serotonin system. J Neurosci. 37(1), 120-128.
- Vincent Beliveau, Brice Ozenne,Stephen Strother, Douglas N. Greve, Claus Svarer, Gitte M. Knudsen, Melanie Ganz (2020) The Structure of the Serotonin System: a PET Imaging Study. NeuroImage. Neuroimage, 205, 116240.
Downloads
Note: Cortical data is mapped in the MNI152 space for visualization purposes only.Bibliography
- Bonaventure P, Hall H, Gommeren W, Cras P, Langlois X, Jurzak M, Leysen JE (2000) Mapping of serotonin 5-HT(4) receptor mRNA and ligand binding sites in the post-mortem human brain. Synapse 36:35-46.
- Comtat C, Sureau FC, Sibomana M, Hong IK, Sjoholm N, Trebossen R (2008) Image based resolution modeling for the HRRT OSEM reconstructions software. In: 2008 IEEE Nuclear Science Symposium Conference Record, pp 4120-4123. IEEE.
- Jovicich J, Czanner S, Greve D, Haley E, van der Kouwe A, Gollub R, Kennedy D, Schmitt F, Brown G, Macfall J, Fischl B, Dale A (2006) Reliability in multi-site structural MRI studies: effects of gradient non-linearity correction on phantom and human data. Neuroimage 30:436-443.
- Knudsen GM et al. (2015) The Center for Integrated Molecular Brain Imaging (Cimbi) Database. Neuroimage:1-7.
- Olesen OV, Sibomana M, Keller SH, Andersen F, Jensen J, Holm S, Svarer C, Højgaard L (2009) Spatial resolution of the HRRT PET scanner using 3D-OSEM PSF reconstruction. IEEE Nucl Sci Symp Conf Rec:3789-3790.
- Sureau FC, Reader AJ, Comtat C, Leroy C, Ribeiro M-J, Buvat I, Trébossen R (2008) Impact of image-space resolution modeling for studies with the high-resolution research tomograph. J Nucl Med 49:1000-1008.
- Varnäs K, Halldin C, Hall H (2004) Autoradiographic distribution of serotonin transporters and receptor subtypes in human brain. Hum Brain Mapp 22:246-260.
- Woods RP, Cherry SR, Mazziotta JC (1992) Rapid automated algorithm for aligning and reslicing PET images. J Comput Assist Tomogr 16:620-633.
Contacts
For questions related to the NRU serotonin atlas, contact Vincent Beliveau (vincent.beliveau-AT-nru.dk) or Melanie Ganz (mganz-AT-nru.dk).Copyright
Copyright (c) 2016, Neurobiology Research Unit, Rigshospital.All rights reserved according to CC 4.0 BY-NC-SA (https://creativecommons.org/licenses/by-nc-sa/4.0).
Redistribution and use in source forms, with or without modification, are permitted provided that the following conditions are met:
* You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.
* You may not use the material for commercial purposes. Neither the name of the Neurobiology Research Unit, Rigshospital nor the names of its contributors may be used to endorse or promote products derived from this image set without specific prior written permission.
* If you remix, transform, or build upon the material, you must distribute your contributions under the same license as the original.